- What Is Life Sciences Software Testing?
- Why Quality Assurance in Life Sciences Industry Is Non-Negotiable
- What Kind of Software Does the Life Sciences Industry Use?
- What Testing Methods are Used for Life Sciences Software?
- Challenges in Life Sciences Software Testing
- Why Should You Adopt Test Automation for the Life Sciences Industry
- Simplify Life Sciences Software Testing with TestGrid
- Final Takeaways for Effective Testing
- Frequently Asked Questions (FAQs)
Life sciences don’t get second chances. A faulty lab software system or an incorrect medical device reading can jeopardize patient safety and invite regulatory actions.
Therefore, you must make sure every algorithm, integration, and interface is flawless and meets strict compliance standards.
But with software systems becoming more complex and interconnected, and patient bases growing, testing every corner of the systems and applications can be tricky.
The good news is, there’s a solution for this issue. A structured life sciences software testing process can help you assess systems under real-world conditions and ensure you release only after every function and feature meets quality benchmarks.
In this blog, we’ll talk in detail about what exactly life sciences software testing is, how you can perform it, the different testing types, and the challenges you might face, along with ways to overcome them.
For scalable, AI-powered life sciences software testing, opt for a free trial with TestGrid.
- Life sciences software testing helps you check the efficiency and reliability of software used in healthcare, pharmaceutical, and research
- Life sciences software includes medical devices, telehealth platforms, EHRs, and health tracking apps
- Testing methods span functional, regression, performance, security, and accessibility checks
- High-impact areas to test are patient record management, imaging systems, insurance claim modules, and remote monitoring platforms
- Best practices to optimize testing efficiency include automation, risk-based validation, continuous monitoring, and comprehensive documentation
What Is Life Sciences Software Testing?
Life sciences software testing is the process of verifying software used in healthcare, medical research, and pharmaceuticals to ensure it meets all functional, regulatory, and safety requirements.
End-to-end testing of life sciences software helps you detect defects, protect sensitive patient data, ensure all modules and integrations work as intended, improve user experience, and release reliable systems that can handle critical healthcare processes.
Why Quality Assurance in Life Sciences Industry Is Non-Negotiable
QA in the life sciences industry isn’t just about verifying code changes. How you test and what you test directly impact a patient’s life.
Imagine if a pacemaker malfunctions. It can be life-threatening.
Even small defects in medical devices and EHR systems can result in incorrect treatment decisions and compliance violations. This is why you must rigorously test every feature, workflow, and data exchange.
A recent report states that cybersecurity failures disrupt patient care in 70% of healthcare organizations. This figure clearly shows how critical robust testing is for apps and software systems in the life sciences sector.
What Kind of Software Does the Life Sciences Industry Use?
The life sciences industry relies on different specialized software systems to enhance operations, improve patient care, and advance scientific research. Some major examples are:
1. Medical devices
Software used in medical devices such as pacemakers, infusion pumps, and glucose monitors helps you in controlling, monitoring, and analyzing their performance.
These software systems ensure that medical devices work correctly and safely. For instance, software in blood glucose monitors reads, processes, and displays blood sugar levels, and also alerts high or low glucose.
2. Telehealth platforms
Telehealth platforms are designed mainly for remote medical consultations via video conferencing or chats. This means patients can get treatment advice from healthcare providers without being in the same physical location.
Telehealth software helps you collect patient information, schedule appointments, and store important medical data.
3. Electronic Health Record (EHR) systems
These are online databases that help you centralize patient records, treatment history, and medical information, and improve patient care coordination.
These software systems collect patient information from healthcare professionals and sync it across departments via secure databases and cloud-based infrastructure.
4. Health tracking apps
The main idea of health and wellness apps is to help individuals monitor their physical activity, diet, sleep patterns, weight, and vital signs.
Basically, these apps integrate wearable sensors such as smartwatches, smartphones, or even manual entries to generate health insights for your users so they can make healthier lifestyle choices.
5. Imaging software
Medical imaging software helps you visualize complex images from diagnostic devices MRI, CT-scan, X-Ray, and ultrasound machines. Radiologists and clinicians normally use these software systems to detect abnormalities, plan treatments, and track disease progress.
6. Online appointment and billing
These software systems take care of the administrative side of healthcare and pharma. They help you automate the process of appointments, billing, insurance claims, and payment processes.
The goal is to carry out financial transactions efficiently, maintain transparency in scheduling, and reduce manual errors.
Also Read: Healthcare Application Testing: The Ultimate Guide to Secure and Reliable Software
What Testing Methods are Used for Life Sciences Software?
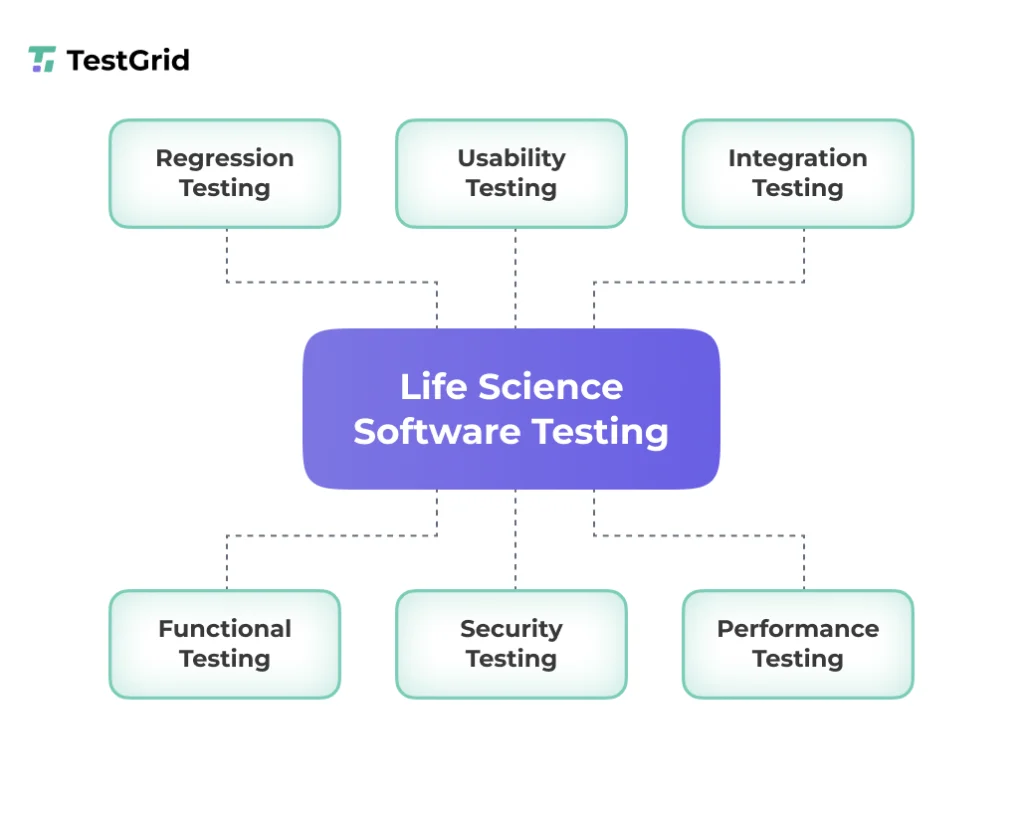
1. Regression testing
Regression testing helps you ensure that new updates or code changes in life sciences software don’t break the existing functionalities or affect performance. This is particularly important for EHRs and medical devices where stability is critical.
Learn More: 13 Regression Testing Tools That Actually Make Testing Easier in 2025
2. Integration testing
Life science software has different components that often interact with each other to deliver quality medical services.
Integration testing allows you to check if different modules such as user authentication, billing, and patient data storage, communicate correctly. You also verify APIs, data formats, and workflows to detect interface errors or mismatches in logic.
3. Usability testing
Life sciences software is mostly used by patients and health professionals like doctors, nurses, labs, and hospitals. And since the software involves critical tasks like entering patient data or preparing diagnostic images, efficiency is vital.
Usability testing helps you check layout, accessibility, and responsiveness to ensure these systems are easy to use so it doesn’t delay treatment.
4. Performance testing
Assessing how well your software handles real-world scenarios including heavy loads and concurrent users is critical to ensure it doesn’t break during clinical use.
Performance testing helps you identify issues such as slow response times or system crashes that can potentially affect patient care.
To check the stability of life sciences software, you can simulate multiple users and high transaction volumes and measure metrics like latency and error rates.
Also Read: Top 20 Performance Testing Tools in 2025
5. Security testing
Life sciences software records sensitive patient information like health records and financial data. And if this data is not stored in a secure way, it can lead to cyberattacks.
Security testing allows you to identify vulnerabilities, including weak authentication, unauthorized access, and data breaches. You can do this by simulating attacks and checking authentication and encryption systems.
6. Functional testing
Functional testing is performed to verify that all functions of the software work as specified in the requirements.
You must thoroughly test features such as patient record management, appointment scheduling, and insurance claim processing to ensure they are foolproof and don’t cause errors during usage.
7. Accessibility testing
Accessibility standards like WCAG 2.1 state that features in software systems such as screen readers, voice commands, keyboard navigation, and color contrast are accessible to all users, including those with disabilities.
Accessibility testing helps you check how well users with visual, auditory, and motor impairments can interact with the apps and other systems.
Check Out: 12 Accessibility Testing Tools That Work in 2025
Challenges in Life Sciences Software Testing
1. Regulatory compliance
Software systems used in the life sciences industry need to strictly comply with standards like HIPAA, GDPR, and ISO 13485. So if there’s even a minor error or missing documentation during testing, it can lead to non-compliance and legal penalties.
| How to overcome You must design a structured process and maintain thorough documentation for all your tests. Make a checklist of the regulatory requirements and take the help of automated compliance testing tools you meet the required standards. |
2. Ensuring data security
Since life sciences software handles sensitive patient and clinical data, protecting it from breaches, unauthorized access, and data corruption can be tough. Complex systems, multiple integrations, and remote access make it even more difficult to manage threats.
| How to overcome When you’re developing the software, implement strong encryption, multi-factor authentication, and role-based access controls. Conduct regular security audits and run continuous penetration tests to identify potential risks early. |
3. Scalability
Life sciences software often faces high numbers of users, large datasets, and transaction volumes. Simulating these real-world load conditions across different systems and medical devices while also maintaining accuracy and speed can be hard.
| How to overcome You can use automated load testing tools and cloud-based environments to scale similar high user loads and scale testing. Make sure the tools you use can adapt seamlessly to growing product lines and user base. |
Why Should You Adopt Test Automation for the Life Sciences Industry
1. Increased efficiency
Automated test scripts allow you to execute repetitive tests like regression, functional, and integration tests much faster compared to manual testing.
With the help of automation tools, you can run these tests across different medical devices, modules, and user scenarios simultaneously. This can speed up feedback and help you release reliable systems faster.
2. Early defect detection and improved coverage
Test automation helps you run continuous tests throughout the development lifecycle. Plus most automation tools integrate easily with your CI/CD workflows.
So testing begins right from the design stage so you can detect issues including edge cases, and fix them early before they escalate and affect your users.
3. Lower cost
Many automation tools allow you to create test scripts that you can reuse for multiple system versions and environments with little to no maintenance. This means you don’t have to validate code changes repeatedly, which lowers operational costs over time.
Simplify Life Sciences Software Testing with TestGrid
TestGrid is an AI-powered end-to-end automation testing platform that helps you test life sciences software with speed, security, and flexibility.
You can generate low-code or no-code automation test scripts and execute them across multiple medical devices in both cloud and on-prem device labs.
The platform offers you secure environments to test your internal apps without exposing endpoints to the public internet. This is particularly helpful for keeping sensitive data and testing in your own network.
With TestGrid’s advanced IoT testing, you can automate end-to-end tests across connected applications and services. The platform simulates data exchanges between components and verifies that your software behaves reliably under real-world usage conditions before release.
Moreover, TestGrid’s enterprise-grade security includes encryption, access controls, 24/7 monitoring, and compliance with standards like GDPR, CCPA, and SOC 2 to help you deploy safe and secure software.
Features like parallel testing, headless browser automation, and real device testing significantly reduce your overall testing time and accelerate release cycles. Plus you can get rich, actionable insights with smart data insights and real-time visual reporting to quickly identify issues and resolve them.
Final Takeaways for Effective Testing
To make the most out of life sciences software testing, automate processes as much as possible to enable continuous testing and reduce the risks of human errors.
Adopt a risk-based approach to prioritize testing areas with high impact, such as patient record management and medical device controls.
Design and maintain clear documentation for audit purposes, train your team to handle complex user scenarios, and implement continuous monitoring and quick feedback loops to find potential issues and resolve them promptly.
And if you want a unified platform that takes care of all your life sciences testing needs and helps you deploy safe and stable software, sign up for a free trial with TestGrid.
Frequently Asked Questions (FAQs)
1. How to perform compliance testing for life sciences software?
You can execute compliance testing with the help of automated tools to verify audit trails, access controls, data integrity, and security features, and check if they meet regulatory standards consistently throughout development and deployment.
2. How do AI-powered tools help in life sciences software testing?
AI-powered tools use predictive analytics to catch potential failures before they happen, self-healing tests to adapt with software changes, and automated compliance monitoring to reduce human error in regulatory processes.
3. What are the regulatory standards in the life sciences industry?
Regulatory standards in the life sciences industry include FDA 21 CFR Part 11 for electronic records, HIPAA for maintaining data privacy, ISO 13485 for medical device quality management, and GxP to ensure medical products are safe and effective.










